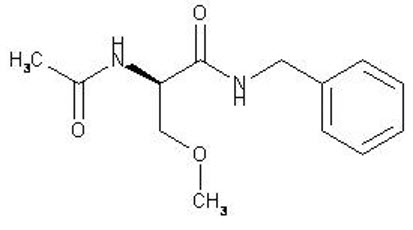
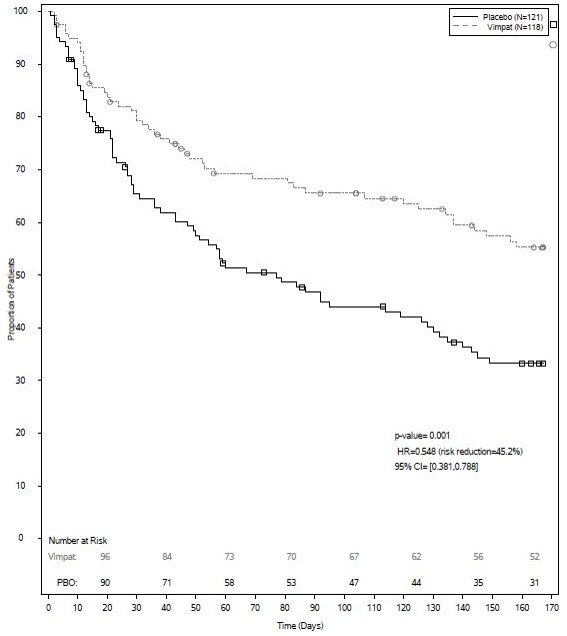
UCB's VIMPAT® (lacosamide) CV now approved by FDA for primary generalized tonic-clonic seizures and expanded pediatric use for people living with epilepsy

Neurological adverse events of new generation sodium blocker antiepileptic drugs. Meta-analysis of randomized, double-blinded studies with eslicarbazepine acetate, lacosamide and oxcarbazepine - ScienceDirect